J. Appl. Cryst. (2010), 43, 1100-1107. Â Â [ doi:10.1107/S0021889810025598 ]
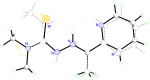
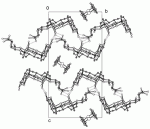
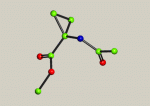
Because they scatter X-rays weakly, H atoms are often abused or neglected during structure refinement. The reasons why the H atoms should be included in the refinement and some of the consequences of mistreatment are discussed along with selected real examples demonstrating some of the … Read the rest