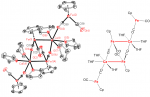
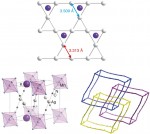
[Update: Thank-you to all the speakers and attendees who made this a great event; details are available on the main Red Kite web page. See you all again next year!]
The inaugural meeting of the Red Kite Crystallographers will be held on Thursday 12th January, 2012 in the Inorganic Chemistry Laboratory, Oxford.
Attendance will … Read the rest