Journal of the American Chemical Society 2016, 138(40), 13314-13325 [doi:10.1021/jacs.6b07501]
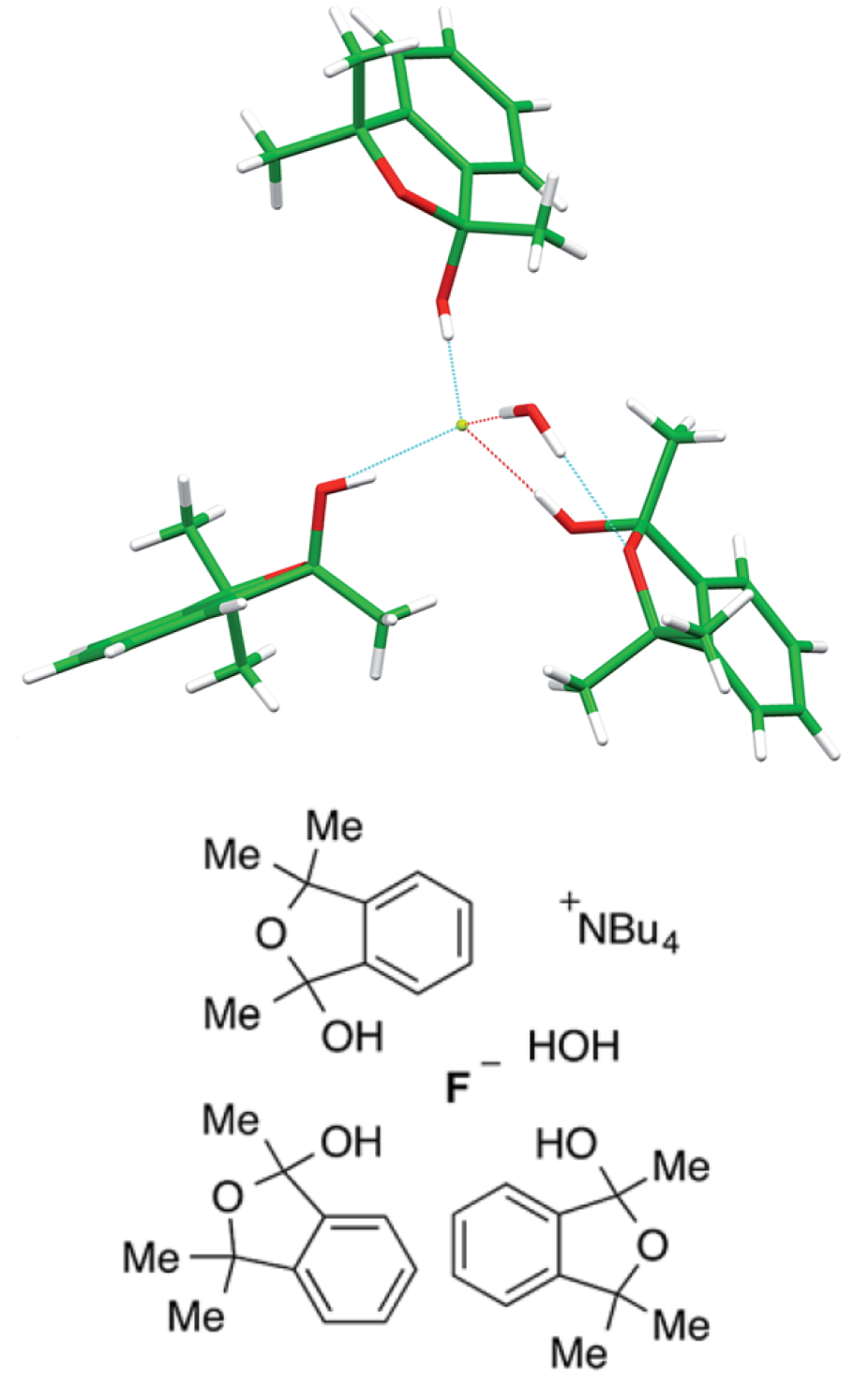
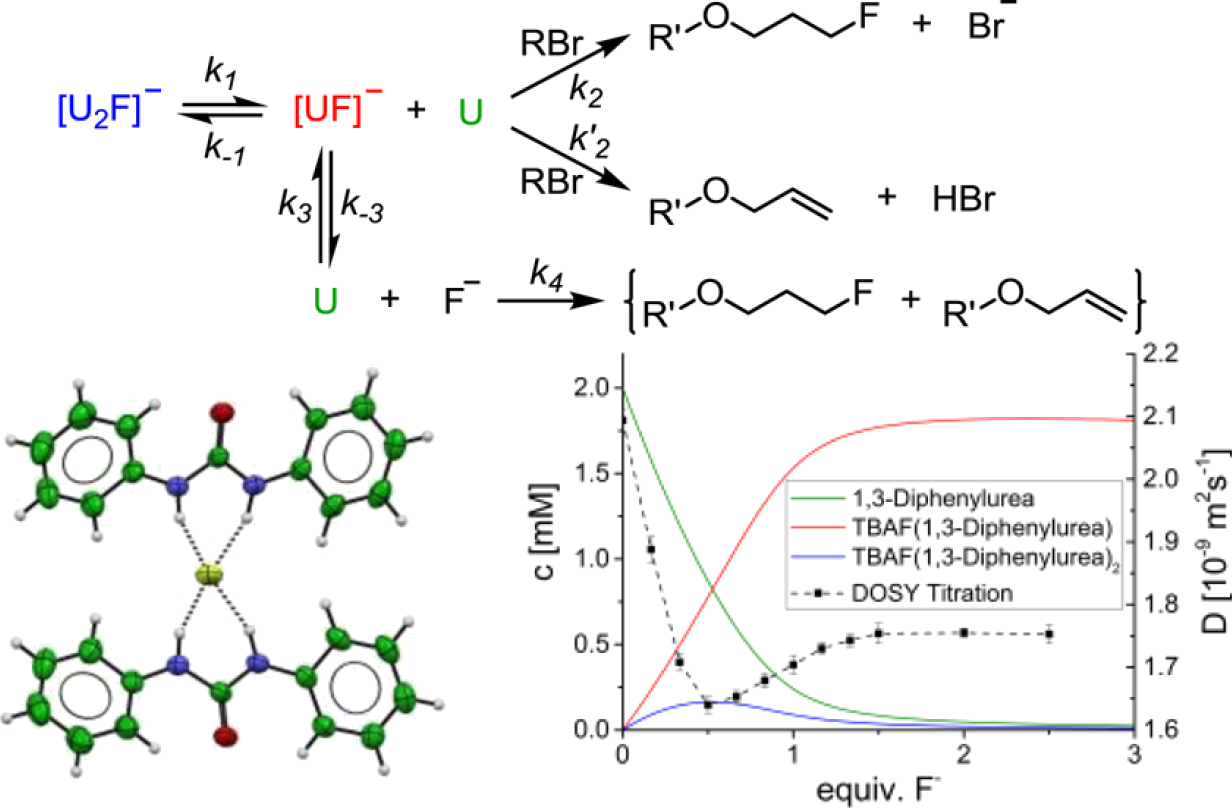
Hydrogen bonding with fluoride is a key interaction encountered when analyzing the mode of action of 5′-ŌĆŗfluoro-ŌĆŗ5′-ŌĆŗdeoxyadenosine synthase, the only known enzyme capable of catalyzing the formation of a C-ŌĆŗF bond from F–ŌĆŗ. Further understanding of the effect of hydrogen bonding on the structure and reactivity of complexed fluoride is therefore important for catalysis and numerous other applications, such as anion supramol. chem. Herein we disclose a detailed study examg. the structure of 18 novel urea-ŌĆŗfluoride complexes in the solid state, by X-ŌĆŗray and neutron diffraction, and in soln. phase and explore the reactivity of these complexes as a fluoride source in SN2 chem. Exptl. data show that the structure, coordination strength, and reactivity of the urea-ŌĆŗfluoride complexes are tunable by modifying substituents on the urea receptor. Hammett anal. of aryl groups on the urea indicates that fluoride binding is dependent on Žāp and Žām parameters with stronger binding being obsd. for electron-ŌĆŗdeficient urea ligands. For the first time, defined urea-ŌĆŗfluoride complexes are used as fluoride-ŌĆŗbinding reagents for the nucleophilic substitution of a model alkyl bromide. The reaction is slower in comparison with known alc.-ŌĆŗfluoride complexes, but SN2 is largely favored over E2, at a ratio surpassing all hydrogen-ŌĆŗbonded complexes documented in the literature for the model alkyl bromide employed. Increased second-ŌĆŗorder rate consts. at higher diln. support the hypothesis that the reactive species is a 1:1 urea-ŌĆŗfluoride complex of type [UF]–ŌĆŗ (U = urea) resulting from partial dissocn. of the parent compd. [U2F]ŌĆŗ–ŌĆŗ. The dissocn. processes can be quantified through a combination of UV and NMR assays, including DOSY and HOESY analyses that illuminate the complexation state and H-ŌĆŗbonding in soln.
Publisher’s copy