Chemical Crystallography: Reflections and Predictions
A symposium to mark the retirement of David J. Watkin
Friday 9th September 2011
[Update: Thanks to all the speakers and everyone who attended this event in honour of DJW.]
Programme
10.00 Â Refreshments in the Lecture Theatre Foyer, Inorganic Chemistry Laboratory
10.50 Â Dr. Richard Cooper (University of Oxford, UK)
Welcome and Introduction
11.10 Â Prof. Howard Flack (University of Geneva, Switzerland)
Working with a Difference
11.40 Â Dr. Matthias Meyer (Agilent Technologies, Poland)
Oxford D(diffraction) – some notes…
12.10 Â Buffet Lunch (University Club, Mansfield Road)
13.40 Â Dr. Alison Edwards (ANSTO, Australia)
Chemical Crystallography with Neutrons: Laue diffraction in the 21st century!
14.10 Â Dr. George Tranter (Chiralabs, Oxford)
Giving Molecules a Hand
14.40 Â Prof. Simon Parsons (University of Edinburgh, UK)
Applications of Leverage Analysis in Chemical Crystallography
15.10 Â Refreshments in the Lecture Theatre Foyer, Inorganic Chemistry Laboratory
15.30 Â Dr. Luc Bourhis (Bruker, Paris)
A Toolbox for Programmer Crystallographers
16.00 Â Prof. Bruce Foxman (Brandeis University, USA)
Secrets from the Oxford Crypt: Old Problems from Keith Prout and John Rollett: Solved!
16.30 Â Dr. Richard Cooper
Closing Remarks
16.45 Â Drinks at the University Club
Many thanks to Agilent Technologies for supporting this meeting. Continue reading »
 Congratulations to Karim Sutton, who has been awarded a Margaret C. Etter Student Lecturer Award at the recent American Crystallographic Association annual meeting in Boston.
Congratulations to Karim Sutton, who has been awarded a Margaret C. Etter Student Lecturer Award at the recent American Crystallographic Association annual meeting in Boston. The
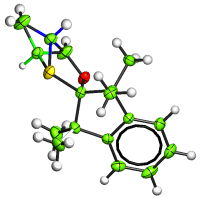
The  Leverages measure the influence that observations (intensity data and restraints) have on the fit obtained in crystal structure refinement. Further analysis enables the influence that observations have on specific parameters to be measured. The results of leverage analyses are discussed in the context of the amino acid alanine and an incomplete high-pressure data set of the complex bis(salicylaldoximato)copper(II). Leverage analysis can reveal situations where weak data are influential and allows an assessment of the influence of restraints. Analysis of the high-pressure refinement of the copper complex shows that the influence of the highest-leverage intensity observations increases when completeness is reduced, but low leverages stay low. The influence of restraints, notably those applying the Hirshfeld rigid-bond criterion, also increases dramatically. In alanine the precision of the Flack parameter is determined by medium-resolution data with moderate intensities. The results of a leverage analysis can be incorporated into a weighting scheme designed to optimize the precision of a selected parameter. This was applied to absolute structure refinement of light-atom crystal structures. The standard uncertainty of the Flack parameter could be reduced to around 0.1 even for a hydrocarbon.
Leverages measure the influence that observations (intensity data and restraints) have on the fit obtained in crystal structure refinement. Further analysis enables the influence that observations have on specific parameters to be measured. The results of leverage analyses are discussed in the context of the amino acid alanine and an incomplete high-pressure data set of the complex bis(salicylaldoximato)copper(II). Leverage analysis can reveal situations where weak data are influential and allows an assessment of the influence of restraints. Analysis of the high-pressure refinement of the copper complex shows that the influence of the highest-leverage intensity observations increases when completeness is reduced, but low leverages stay low. The influence of restraints, notably those applying the Hirshfeld rigid-bond criterion, also increases dramatically. In alanine the precision of the Flack parameter is determined by medium-resolution data with moderate intensities. The results of a leverage analysis can be incorporated into a weighting scheme designed to optimize the precision of a selected parameter. This was applied to absolute structure refinement of light-atom crystal structures. The standard uncertainty of the Flack parameter could be reduced to around 0.1 even for a hydrocarbon.