Presented by:Â Nicholas H. Evans & Christopher J. Serpell
Research Leader:Â Prof. Paul D. Beer
Published: Angewandte Chemie International Edition
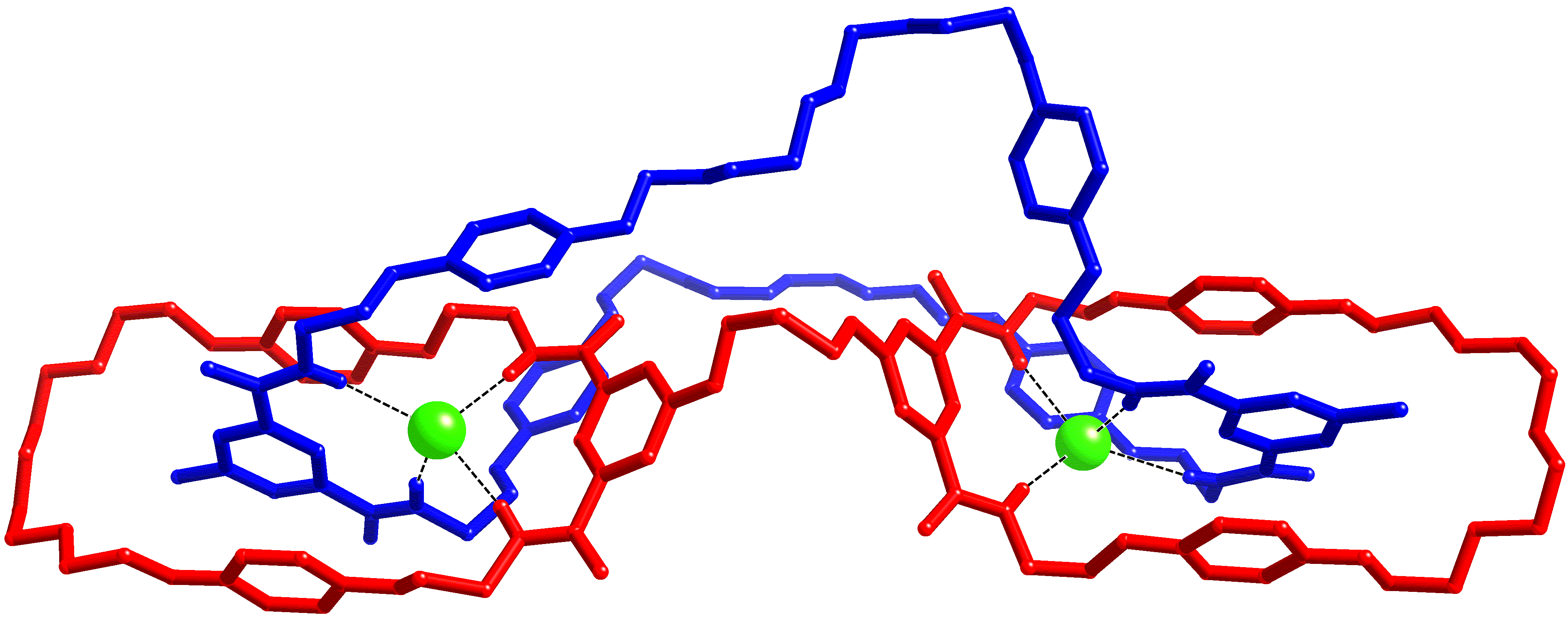
Catenanes and rotaxanes are highly attractive targets for the supramolecular chemist due to their potential uses as molecular machines or as selective hosts for ionic and molecular guests. This molecule was synthesised via chloride anion templation and crystals grown by slow diffusion of diisopropyl ether into a chloroform/catenane solution. Data were collected on I19 at Diamond.  This first handcuff catenane structure provided proof of the topology, also revealing potential further uses: the degree of slack in the large macrocycle could allow controlled rotation within the handcuff, and the large number of oxygen atoms in the cavity formed by the handcuff linker and large macrocycle could be used to bind cations.